Spin particles
Particles are defined with the following equation
particle = incidence * symmetry * nuclei
where incidence is the incidence of the particle, symmetry is the two-fold symmetry (1 or 2) and the nuclei is number of spins.
The table below shows some examples for the particles.
| Compound | Particle | Spin System |
|---|---|---|
| one proton | 1*1*1 | |
| 1,2-dicloro benzene | 1*2*1 | AA’BB’ |
| CH2 in ethanol | 1*1*2 | A2 |
| CH3 in ethanol | 1*1*3 | A3 |
| 2*CH3 in dimethyl benzene | 1*2*3 | A3A3’ |
| TSP | 3*1*3 (or 9*1*1) | |
| Triphenylmethane | 1*1*1, 3*2*1, 3*2*1, 3*1*1 |
The following examples include atom assignments for chemical shifts:
- A3BBCCDD_Butylamine
- AABBC_Benzoic_acid
- AABBC_Phenol
- AABBCR2X_Hippuric_acid
- AABBR2X_4OH-Hippuric_acid
Defining spin particles for chemical shifts
After the new spin systems has been created, you need to define the spin particles for the chemical shifts. The particles can be edited by double clicking the cells in the Shifts panel (see the picture below).
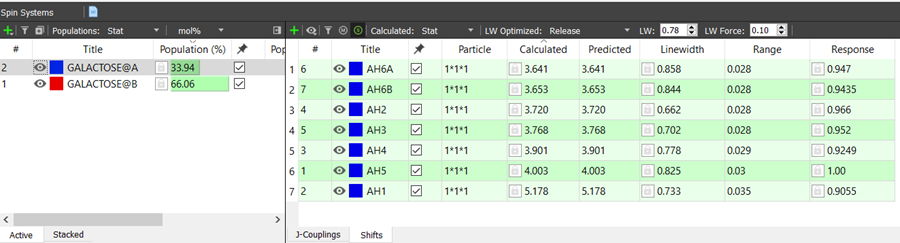
The particles can be viewed on the spectrum window by selecting Shift Labels → Particle:

The assignments can be highlighted by hovering the mouse over the chemical shift labels.

Tips
- Aromatic rings:
- Orto: 8 Hz;
- Meta: 2 Hz;
- Para: 1 Hz;
- Pyridine:

- 5-membered sp2-rings:
- Aliphatic 3J = 1 - 15 Hz, depends on substituents and dihedral angle between the protons. If not known and the spectrum is simple, 8 Hz is a fair guess.
- For 3JAB and 3JAB' (=3JBA) of AA'BB'-system formed by -CH2-CH2- use different values, for example, 6 and 8 Hz.
- 2J(sp3) = -14 Hz (CH2 in aliphatic structure), depends on substituent electronegativity (range from -8 to -18 Hz). 2J(sp2) = 2 Hz (in C=CH2).
- Check that the sign of 2J(sp3) is negative!
- Cyclic compounds may also need:
- 4J > 2 Hz for W-shaped pathway, otherwise negative (>-0.5 Hz) and often negligible;
- 5J > 0 Hz for W-shaped pathway, otherwise negligible;
- 6J < 0 Hz for W-shaped pathway, usually negligible.
- Substituent α-protons couple to aromatic ring protons with 4J = 0 to -0.5 Hz, 5J = 0 to 0.75 Hz, 6J = 0 to -1.2 Hz, which depend on α-proton-ring dihedral angle. These couplings can be often ignored – their contribution is then included into line-widths.
J-couplings
The couplings should be checked before proceeding to the spin system optimization. As the initial parameters are predicted, they may lack for example long-range couplings, or the signs of the 2J-couplings might be wrong.
Here are some examples for initial values of couplings:

ABCD and AA’BB’ systems
AA’BB’ system may have been given as an ABCD system and it is recommended that it is packed into AA’BB’ system. ABCD-system could work but only when shifts are equalized and fixed and couplings are equalized.